1.вЧЦїМђНщ
TIC-600ЪЧluck18ЙйЭјвЧЦїШЋаФбаЗЂЕФШЋЫмЛЏЭЈгУаЭЁЂзддйЩњвжжЦФЃЪНЕФРызгЩЋЦзвЧ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌПЩгУгкМьВтЭЈР§ЮоЛњвѕбєРызгЁЂвћгУЫЎЯћЖОИБВњЦЗЁЂфхЫсбЮЁЂгаЛњЫсЁЂАЗЕШ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌВЛЕЋгЕгаЦеБщЁЂЭъЩЦЁЂЯШНјЕФгІгУНтОіФмСІ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЭЌЪБЮЊгУЛЇДјРДздЖЏЛЏЁЂШЫадЛЏЕФвЧЦїгІгУЬхбщ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌПЩЦеБщгІгУгкЧщаЮЁЂЪГЮяЁЂЛЏЙЄЁЂХЉвЕЁЂПѓВњЁЂЕчзгЕчЦїЕШаавЕжаГЃСПМАЮЂКлСПРызгЕФМьВтЦЪЮіЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ

ЭМ1. TIC-600РызгЩЋЦзвЧЭтЙлЭМ
2.РызгЩЋЦздкЫЎжЪМьВтжаЕФгІгУ
ТШЫсбЮЁЂбЧТШЫсбЮЁЂфхЫсбЮЁЂЖўТШввЫсКЭШ§ТШввЫсЪЧГЃМћЯћЖОИБВњЦЗЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЩњбФЮлЫЎМАЙЄвЕЗЯЫЎЃЈШчвНСЦЗЯЫЎЃЉашвЊЭЖМгИќИпМССПЕФЯћЖОМС�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌвдЕжДяХХЗХКЭЛигУБъзМ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖддйЩњЫЎМАЕиБэЫЎЕФЧхОВЪЙгУдьГЩЧБдкЕФЮЃЯе�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌНјЖјМгжиЫЎЬхЕФЮлШОЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃГЃгУЕФЯћЖОМСгавКТШЁЂЖўбѕЛЏТШЁЂДЮТШЫсФЦЁЂзЯЭтЯпЁЂТШАЗКЭГєбѕЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
ТШЫсбЮКЭбЧТШЫсбЮ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌжївЊЪЧаТаЭЯћЖОМСЖўбѕЛЏТШЕФЯћЖОИБВњЦЗ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЦфЖОРэбЇгАЯьНЯДѓ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌбЧТШЫсбЮФмв§Ц№ЖЏЮяЕФШмбЊадбЊащКЭБфадбЊКьТбАзбЊАЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌвжжЦбЊЧхМззДЯйЫиЕФзїгУ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌв§Ц№ЬЅЖљаЁФджиСПЯТНЕЁЂЩёОааЮЊзїгУЛКТ§ЛђЯИАћЪ§ЯТНЕЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃТШЫсбЮЪЧЩёОЁЂаФбЊЙмКЭКєЮќЕРжаЖОгыМззДЯйЫ№КІбЊащЕФгевђжЎвЛ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЦфЖОадЛсНЕЕЭОЋзгЕФЪ§ФПКЭЛюСІЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃШЫЬхЦЄЗєНгДЅЛђЮќШыТШЫсМиКѓ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЛсЕМжТКєЮќЯЕЭГМВВЁЗКЦ№аФЗГЁЂЭТФцЁЂИЙаКЁЂЦЄЗєЙ§УєЕШжЂзД�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌбЯжиЪБЛсЗКЦ№ШмбЊЁЂЛЦЮЎВЁЁЂФђЖОжЂЁЂГщДЄЁЂЛшиЪжБжСИЮЩіЙІаЇЫЅНпЖјЫР�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌКуОУНгДЅТШЫсМи�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌПЩв§Ц№ЪГгћВЛеёЁЂЬхжиЯТНЕ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЩѕжСгеЗЂАЉжЂ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖдГЩШЫЕФжТЫРСПЮЊ 12 gЁЂЖљЭЏЮЊ 5 gЁЂгЄЖљЮЊ 1
gЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃжВЮяЮќЪеClO3-ЛсвжжЦжВЮяЯИАћЖд NO3-ЕФЮќЪеКЭдЫЪф�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЕМжТжВЮяШБЕЊ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌНјЖјгАЯьжВЮяЬхЕФаФРэЁЂгЊбјКЭЩњжГЩњГЄЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
фхЫсбЮжївЊЪЧвдГєбѕзїЮЊЯћЖОМСДІжУГЭЗЃКЌфхЕФЫЎЫљЬьЩњЕФИБВњЦЗЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃШєдЫЎжаКЌга Br-, Br-�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌОЭЛсБЛГєбѕЗжзгжБНгбѕЛЏЬьЩњфхЫсбЮ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЛђепЭЈЙ§-OH бѕЛЏЬьЩњфхЫсбЮЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃфхЫсбЮПЩдьГЩЫЎЩњЩњЮяЃЈШчДѓаЭдщЁЂТуИЙдщЁЂАпТэгуЕШЃЉЩњГЄЫйТЪБфТ§ЁЂдЫЖЏЪмвжжЦЛђщцУќТЪдіЬэЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃвђЦфПЩгеЗЂЪдбщЖЏЮяЩідрЯИАћжзСі�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЧвгавХДЋЖОад�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЙЪфхЫсбЮБЛЙњМЪАЉжЂбаОПЛњЙЙЖЈЮЊ 2B МЖ(НЯИпжТАЉПЩФмад)ЕФЧБдкжТАЉЮяЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃвЛИіЬхжи 70 kg ЕФГЩФъШЫ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЬьЬьвћЫЎ 2 L�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЕБфхЫсбЮХЈЖШЮЊ45 ІЬg/LЁЂ0.5 ІЬg/L КЭ 0.05 ІЬg/L ЪБ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌдђжеЩэжТАЉТЪЮЊ 10-4ЁЂ10-5 КЭ 10-6�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌжТАЉЮЃКІНЯДѓЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
ЖўТШввЫсЃЈDCAAЃЉКЭШ§ТШввЫсЃЈTCAAЃЉжївЊЪЧТШЛЏЯћЖОИБВњЦЗжаТБввЫсРрЕФДњБэЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃTCAAОпгажТЛћзїгУ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌDCAA вВБЛУРЙњЧщаЮБ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛЄОжЕФЧхОВЫЎЗЈСаЮЊПЩвЩжТАЉЮяЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃТБввЫсвбБЛжЄЪЕЖдрПГнРрЖЏЮягажТАЉЁЂжТЛћБфЁЂжТЭЛБфзїгУ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌжТАЉЮЃКІДѓДѓИпгкЦфЫќЯћЖОИБВњЦЗЕФзмКЭ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌгабаОПХњзЂ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌТБввЫсЕФжТАЉЮЃКІеМЯћЖОИБВњЦЗзмжТАЉЮЃКІЕФ 91.9%вдЩЯЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
ФПНёвћгУЫЎжЪСПЮЪЬтдНРДдНЪмжиЪг�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌИїЕивВЪБЪББЌГіЫЎжЪЮлШОЮЪЬт�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЪЙЕУеўИЎВПЗжМАИїЕиЫЎжЪМрПиЁЂздРДЫЎЙЉгІГЇдНРДдНжиЪгЫЎжЪМьВт�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖјвћгУЫЎжаBr-ЁЂBrO3-ЁЂClO2-ЁЂClO3-ЁЂЖўТШввЫсЁЂШ§ТШввЫсЕШвѕРызгКЌСПЕФМИЖр�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌжБНггАЯьздРДЫЎЫЎжЪ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌвбГЩЮЊЫЎжЪМьВтЕФЭЈР§ПЦФПЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
РызгЩЋЦзвЧTIC-600вђОпгаМьГіЯоЕЭЁЂзМШЗЖШИпЁЂОМУЪЪгУЁЂВйзїОЋСЗРћБуЕШгХЕу�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌПЩЦеБщгІгУгквћгУЫЎжавѕРызгКЌСПЕФМьВтЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
3.гІгУЪЕР§
3.1 РызгЩЋЦзЗЈВтЖЈЫЎжаBr-ЁЂClO2-ЁЂClO3-Ш§жжвѕРызг
3.1.1 ВЮПМБъзМ GB/T5750.10-2006ЃЈЩњбФвћгУЫЎжаBr-ЁЂClO2-ЁЂClO3-ЕФВтЖЈЃЉ
3.1.2 ЪЪгУЙцФЃ ЪЪгУгкЩњбФвћгУЫЎМАЫЎдДжабЧТШЫсбЮЁЂТШЫсбЮЁЂфхРызгЕФВтЖЈ
3.1.3ЪЕбщВПЗж
3.1.3.1ЪдМСМАвЧЦї
СмЯДвКЃКЬМЫсЧтФЦ[C(NaHCO3)=1.7mmol/l]-ЬМЫсФЦ[C(Na2CO3)=1.6mmol/l]ЃКГЦШЁ0.1428gЬМЫсЧтФЦКЭ0.1908gЬМЫсФЦ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШмгкГЌДПЫЎВЂЯЁЪЭЕН1LЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЃЈЙњвЉМЏЭХЛЏбЇЪдМСгаЯоЙЋЫОЃЉ
фхЛЏЮяБъвКЃЈ1000ІЬg/mlЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌТШЫсбЮБъвКЃЈ1000ІЬg/mlЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌбЧТШЫсбЮБъвКЃЈ1000ІЬg/mlЃЉЃЈЙњМвБъзМЮяжЪжааФЃЉ
РызгЩЋЦзвЧЃЈTIC-600�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌluck18ЙйЭјЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌBS 224SЕчзгЬьЦНЃЈШќЖрРћЫЙПЦбЇвЧЦїЃЈББОЉЃЉгаЯоЙЋЫО�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ0.1mgЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛSHZ-DаЭбЛЗЫЎЪНецПеБУЃЈЙЎвхЪагшЛЊвЧЦїгаЯоЙЋЫОЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛвЦвКЧЙЃЈЕТЙњЦеРМЕТ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ10-100uLЁЂ100-1000uLЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛ0.22ІЬmЮЂПзТЫФЄЃЈЩЯКЃАВЦзЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШмМСГщТЫзАжУЃЈНђЬкЪЕбщзАБИгаЯоЙЋЫОЃЉ
ЪдбщЫљгУЫЎОљЮЊГЌДПЫЎЃЈЕчзшТЪДя18.25MІИЁЄcmЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЫљгУЦїУѓОљгУГЌДПЫЎЯДхЊВЂНўХнзЁЫоЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЃЈA-10ГЌДПЫЎЛњ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌУРЙњУмРэВЉЃЉ
3.1.3.2 ЪЕбщРњГЬ
бљЦЗдЄДІжУГЭЗЃЃК
НЋЫЎбљО0.22ІЬmЮЂПзТЫФЄЙ§ТЫГ§ЮлзЧдгжЪ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖдгВЖШИпЕФЫЎбљ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌаывЊЪБПЩОбєРызгНЛСїЪїжЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌдйО0.22ІЬmЮЂПзТЫФЄЙ§ТЫ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖдКЌгаЛњЮяЕФКгЫЎЕШЕиБэЫЎбљ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌПЩЯШОC18жљЙ§ТЫГ§ШЅЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
бљЦЗОдЄДІжУГЭЗЃКѓзЂШыРызгЩЋЦзвЧНјбљЯЕЭГ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌМЭТМЗхИпЛђЗхУцЛ§ЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
БъвКХфжЦЃК
ЛЎЗжвЦШЁвЛЖЈСПЕФфхЛЏЮяБъвКЃЈ1000ІЬg/mlЃЉЁЂбЧТШЫсбЮБъвКЃЈ1000ІЬg/mlЃЉЁЂ
ТШЫсбЮБъвКЃЈ1000ІЬg/mlЃЉжС100mlВЃСЇШнСПЦП�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЩшжУГЩХЈЖШШчЯТБэ1ЫљЪОЯЕСаЛьЯ§БъвКЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
Бэ1.ЛьЯ§БъвКХЈЖШ
|
вѕРызг
|
БъвКХЈЖШЃЈІЬg/lЃЉ
|
|
Std1
|
Std2
|
Std3
|
Std4
|
|
Br-
|
50
|
100
|
200
|
400
|
|
ClO2-
|
50
|
100
|
200
|
400
|
|
ClO3-
|
50
|
100
|
200
|
400
|
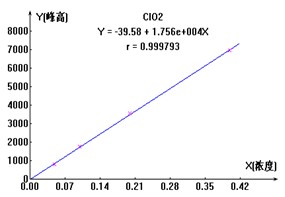
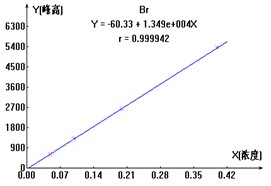
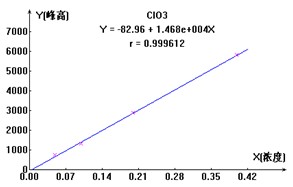
ЭМ2. ClO2-ЁЂBr-ЁЂClO3-Ш§жжвѕРызгБъзМЧњЯпЭМ
3.1.3.3ЪЕбщЪ§Он
1. бљЦЗМгБъНгФЩТЪ
НЋздРДЫЎбљЦЗЛЎЗжМгБъВюБ№ХЈЖШ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШЛКѓНЋУПЗнбљЦЗЕФбљЦЗШмвКНјбљВтЖЈ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЦОжЄВтЪдаЇЙћХЬЫуМгБъНгФЩТЪ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЯъЯИЪ§ОнМћБэ2ЁЂБэ3ЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
Бэ2.здРДЫЎбљЦЗМгБъНгФЩТЪЪ§Он
|
вѕРызг
|
ВтЪдаЇЙћ/ІЬg/ml
|
|
здРДЫЎ
|
МгБъ50 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ100 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ200 ІЬg/l
|
НгФЩТЪ/%
|
|
ClO2-
|
0
|
53.4
|
106.8
|
100.9
|
100.9
|
193.8
|
96.9
|
|
Br-
|
68.6
|
118.4
|
99.7
|
169.4
|
100.8
|
274.1
|
102.8
|
|
ClO3-
|
30.9
|
80.0
|
98.2
|
133.8
|
102.9
|
234.7
|
101.9
|
Бэ3.КгЫЎбљЦЗМгБъНгФЩТЪЪ§Он
|
вѕРызг
|
ВтЪдаЇЙћ/ІЬg/ml
|
|
здРДЫЎ
|
МгБъ50 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ100 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ200 ІЬg/l
|
НгФЩТЪ/%
|
|
ClO2-
|
0
|
45.7
|
91.4
|
92.5
|
92.5
|
195.9
|
97.9
|
|
Br-
|
0
|
50.2
|
100.4
|
102.6
|
102.6
|
200.4
|
100.2
|
|
ClO3-
|
126.9
|
177.8
|
101.8
|
224.2
|
97.3
|
328.7
|
100.9
|
3.1.4НсТл
гЩЪ§ОнПЩжЊ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌРызгЩЋЦзЗЈВтЪдЫЎжаBr-ЁЂClO2-ЁЂClO3-Ш§жжвѕРызгзМШЗЖШИп�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌзюаЁМьГіХЈЖШЕЭ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌвЊСьМђЦгПьНн�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЭъШЋжЊзуЛЗБЃаавЕЙигкЫЎжаBr-ЁЂClO2-ЁЂClO3-Ш§жжвѕРызгМьВтЕФашЧѓЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
3.2 РызгЩЋЦзЗЈВтЖЈЫЎжаBrO3-Рызг
3.2.1ВЮПМБъзМ GB/T5750.10-2006ЃЈЩњбФвћгУЫЎжафхЫсбЮЕФВтЖЈЃЉ
3.2.2 ЪЪгУЙцФЃ ЪЪгУгкЩњбФвћгУЫЎМАЫЎдДжабЧТШЫсбЮЁЂТШЫсбЮЁЂфхРызгЕФВтЖЈ
3.2.3ЪЕбщВПЗж
3.2.3.1 ЪдМСМАвЧЦї
СмЯДвКЃКЬМЫсЧтФЦ[C(NaHCO3)=1.7mmol/l]-ЬМЫсФЦ[C(Na2CO3)=1.6mmol/l]ЃКГЦШЁ0.1428gЬМЫсЧтФЦКЭ0.1908gЬМЫсФЦ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШмгкГЌДПЫЎВЂЯЁЪЭЕН1LЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЃЈЙњвЉМЏЭХЛЏбЇЪдМСгаЯоЙЋЫОЃЉ
фхЫсбЮБъвКЃЈ1000ІЬg/mlЃЉЃЈЙњМвБъзМЮяжЪжааФЃЉ
РызгЩЋЦзвЧЃЈTIC-600�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌluck18ЙйЭјЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌBS
224SЕчзгЬьЦНЃЈШќЖрРћЫЙПЦбЇвЧЦїЃЈББОЉЃЉгаЯоЙЋЫО�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ0.1mgЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛSHZ-DаЭбЛЗЫЎЪНецПеБУЃЈЙЎвхЪагшЛЊвЧЦїгаЯоЙЋЫОЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛвЦвКЧЙЃЈЕТЙњЦеРМЕТ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ10-100uLЁЂ100-1000uLЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛ0.22ІЬmЮЂПзТЫФЄЃЈЩЯКЃАВЦзЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШмМСГщТЫзАжУЃЈНђЬкЪЕбщзАБИгаЯоЙЋЫОЃЉ
ЪдбщЫљгУЫЎОљЮЊГЌДПЫЎЃЈЕчзшТЪДя18.25MІИЁЄcmЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЫљгУЦїУѓОљгУГЌДПЫЎЯДхЊВЂНўХнзЁЫоЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЃЈA-10ГЌДПЫЎЛњ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌУРЙњУмРэВЉЃЉ
3.2.3.2 ЪЕбщРњГЬ
бљЦЗдЄДІжУГЭЗЃЃК
НЋЫЎбљО0.22ІЬmЮЂПзТЫФЄЙ§ТЫГ§ЮлзЧдгжЪ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖдгВЖШИпЕФЫЎбљ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌаывЊЪБПЩОбєРызгНЛСїЪїжЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌдйО0.22ІЬmЮЂПзТЫФЄЙ§ТЫ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖдКЌгаЛњЮяЕФКгЫЎЕШЕиБэЫЎбљ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌПЩЯШОC18жљЙ§ТЫГ§ШЅЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЫЎжаТШРызгЖдBrO3-РызгВтЖЈБЌЗЂгАЯь�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌбљЦЗашОЙЬЯрнЭШЁжљAgжљЙ§жљДІжУГЭЗЃвдГ§ШЅClРызгЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
бљЦЗОдЄДІжУГЭЗЃКѓзЂШыРызгЩЋЦзвЧНјбљЯЕЭГ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌМЭТМЗхИпЛђЗхУцЛ§ЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
БъвКХфжЦЃК
вЦШЁвЛЖЈСПЕФфхЫсбЮБъвКЃЈ1000ІЬg/mlЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЩшжУГЩХЈЖШЮЊ10ІЬg/mlЕФфхЫсбЮДЂБИвК�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌдйЛЎЗжвЦШЁ0mlЁЂ0.1mlЁЂ0.2mlЁЂ0.5mlЁЂ1.0mlЕФ10ІЬg/mlЕФфхЫсбЮДЂБИвКжС100mlВЃСЇШнСПЦП�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌВЂМгГЌДПЫЎЖЈШн�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌХфжЦГЩ0ІЬg/lЁЂ10ІЬg/lЁЂ20ІЬg/lЁЂ50ІЬg/lЁЂ100ІЬg/lЕФЯЕСаБъзМШмвК�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШчЯТЭМ3 ЮЊBrO3-РызгБъзМЧњЯпЭМЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
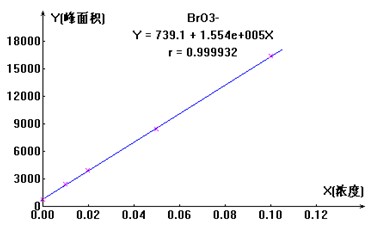
ЭМ3. BrO3-РызгБъзМЧњЯпЭМ
3.2.3.3ЪЕбщЪ§Он
1.бљЦЗМгБъНгФЩТЪ
НЋздРДЫЎбљЦЗЁЂКгЫЎбљЦЗЛЎЗжМгБъВюБ№ХЈЖШ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШЛКѓНЋУПЗнбљЦЗЕФбљЦЗШмвКНјбљВтЖЈ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШЛКѓЦОжЄВтЪдаЇЙћХЬЫуМгБъНгФЩТЪ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЯъЯИЪ§ОнМћБэ4ЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
Бэ4. здРДЫЎМАКгЫЎбљЦЗМгБъНгФЩТЪЪ§Он
|
бљЦЗ
|
BrO3-ВтЪдаЇЙћ/ІЬg/l
|
|
ЮДМгБъ
|
МгБъ10 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ20 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ40 ІЬg/l
|
НгФЩТЪ/%
|
|
здРДЫЎ
|
1.1
|
10.8
|
97.0
|
20.48
|
96.9
|
38.7
|
96.8
|
|
КгЫЎ
|
0
|
10.41
|
104.1
|
21.33
|
106.7
|
38.8
|
97.0
|
3.2.4НсТл
гЩЪЕбщЪ§ОнПЩжЊ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌРызгЩЋЦзЗЈВтЪдЫЎжаBrO3-вѕРызгзМШЗЖШИп�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌМьГіХЈЖШЕЭ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌвЊСьМђЦгПьНн�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЭъШЋжЊзуЛЗБЃаавЕЙигкЫЎжаBrO3-РызгМьВтЕФашЧѓЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
3.3 РызгЩЋЦзЗЈВтЖЈЫЎжаЖўТШввЫсЁЂШ§ТШввЫс
3.3.1ВЮПМБъзМ GB/T5749-2006ЃЈЩњбФвћгУЫЎЮРЩњБъзМЃЉ
3.3.2 ЪЪгУЙцФЃ ЪЪгУгкЩњбФвћгУЫЎМАЫЎдДжаЖўТШввЫсЁЂШ§ТШввЫсЕФВтЖЈ
3.3.3ЪЕбщВПЗж
3.3.3.1 ЪдМСМАвЧЦї
СмЯДвКЃКЬМЫсЧтФЦ[C(NaHCO3)=3mmol/l]-ЬМЫсФЦ[C(Na2CO3)=3mmol/l]ЃКГЦШЁ0.252gЬМЫсЧтФЦКЭ0.3018gЬМЫсФЦ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШмгкГЌДПЫЎВЂЯЁЪЭЕН1LЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЃЈЙњвЉМЏЭХЛЏбЇЪдМСгаЯоЙЋЫОГЇМвЃЉ
ЖўТШввЫсЁЂШ§ТШввЫсБъвКЃЈ1000ІЬg/mlЃЉЃЈЙњМвБъзМЮяжЪжааФЃЉ
РызгЩЋЦзвЧЃЈTIC-600�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌluck18ЙйЭјЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌBS
224SЕчзгЬьЦНЃЈШќЖрРћЫЙПЦбЇвЧЦїЃЈББОЉЃЉгаЯоЙЋЫО�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ0.1mgЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛSHZ-DаЭбЛЗЫЎЪНецПеБУЃЈЙЎвхЪагшЛЊвЧЦїгаЯоЙЋЫОЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛвЦвКЧЙЃЈЕТЙњЦеРМЕТ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ10-100uLЁЂ100-1000uLЃЉ�ЃЛ�ЃЛ�ЃЛ�ЃЛ�ЃЛ0.22ІЬmЮЂПзТЫФЄЃЈЩЯКЃАВЦзЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШмМСГщТЫзАжУЃЈНђЬкЪЕбщзАБИгаЯоЙЋЫОЃЉ
ЪдбщЫљгУЫЎОљЮЊГЌДПЫЎЃЈЕчзшТЪДя18.25MІИЁЄcmЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЫљгУЦїУѓОљгУГЌДПЫЎЯДхЊВЂНўХнзЁЫоЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃЃЈA-10ГЌДПЫЎЛњ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌУРЙњУмРэВЉЃЉ
3.3.3.2 ЪЕбщРњГЬ
бљЦЗдЄДІжУГЭЗЃЃК
НЋЫЎбљО0.22ІЬmЮЂПзТЫФЄЙ§ТЫГ§ЮлзЧдгжЪ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖдгВЖШИпЕФЫЎбљ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌаывЊЪБПЩОбєРызгНЛСїЪїжЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌдйО0.22ІЬmЮЂПзТЫФЄЙ§ТЫ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЖдКЌгаЛњЮяЕФКгЫЎЕШЕиБэЫЎбљ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌПЩЯШОC18жљЙ§ТЫГ§ШЅЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃСэЫЎжаТШРызгЖдЖўТШввЫсРызгВтЖЈБЌЗЂгАЯь�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌбљЦЗашОЙЬЯрнЭШЁжљAgжљЙ§жљДІжУГЭЗЃвдГ§ШЅClРызгЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
бљЦЗОдЄДІжУГЭЗЃКѓзЂШыРызгЩЋЦзвЧНјбљЯЕЭГ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌМЭТМЗхИпЛђЗхУцЛ§ЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
БъвКХфжЦЃК
вЦШЁвЛЖЈСПЕФЖўТШввЫсБъвКЃЈ1000ІЬg/mlЃЉЁЂШ§ТШввЫсБъвКЃЈ1000ІЬg/mlЃЉ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЩшжУГЩЕФЖўТШввЫсЃЈХЈЖШЮЊ10ІЬg/mlЃЉЁЂШ§ТШввЫсЃЈХЈЖШЮЊ5.0ІЬg/mlЃЉЛьЯ§ДЂБИвК�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌдйЛЎЗжвЦШЁ0mlЁЂ1.0mlЁЂ2.0mlЁЂ5.0mlЁЂ10.0mlЕФ10ІЬg/mlЕФЖўТШввЫсЁЂШ§ТШввЫсЛьЯ§ДЂБИвКжС100mlВЃСЇШнСПЦП�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌВЂМгГЌДПЫЎЖЈШн�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌХфжЦГЩЖўТШввЫсЁЂШ§ТШввЫсЯЕСаЛьЯ§БъзМШмвК�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШчЯТЭМ4 ЮЊЖўТШввЫсЁЂШ§ТШввЫсРызгБъзМЧњЯпЭМЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
Бэ5.ЖўТШввЫсЁЂШ§ТШввЫсЛьЯ§БъвКХЈЖШ
|
вѕРызг
|
БъвКХЈЖШЃЈІЬg/lЃЉ
|
|
Std1
|
Std2
|
Std3
|
Std4
|
|
ЖўТШввЫс
|
50
|
100
|
200
|
500
|
|
Ш§ТШввЫс
|
100
|
200
|
400
|
1000
|
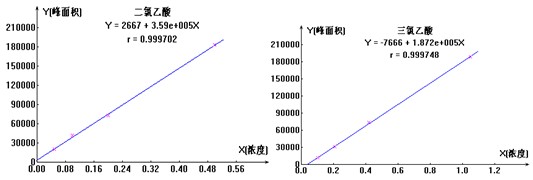
ЭМ4. ЖўТШввЫсЁЂШ§ТШввЫсБъзМЧњЯпЭМ
3.3.3.3ЪЕбщЪ§Он
1.бљЦЗМгБъНгФЩТЪ
НЋздРДЫЎбљЦЗЁЂКгЫЎбљЦЗЛЎЗжМгБъВюБ№ХЈЖШЖўТШввЫсЁЂШ§ТШввЫс�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШЛКѓНЋУПЗнбљЦЗЕФбљЦЗШмвКНјбљВтЖЈ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌШЛКѓЦОжЄВтЪдаЇЙћХЬЫуМгБъНгФЩТЪ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЯъЯИЪ§ОнМћБэ6ЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ
Бэ6. здРДЫЎМАКгЫЎбљЦЗМгБъНгФЩТЪЪ§Он
|
бљЦЗ
|
ЖўТШввЫсВтЪдаЇЙћ/ІЬg/l
|
|
ЮДМгБъ
|
МгБъ100 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ200 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ500ІЬg/l
|
НгФЩТЪ/%
|
|
здРДЫЎ
|
0
|
91.3
|
91.3
|
196.3
|
98.2
|
486.2
|
97.3
|
|
КгЫЎ
|
0
|
94.2
|
94.2
|
192.1
|
96.1
|
503.2
|
100.6
|
|
бљЦЗ
|
Ш§ТШввЫсВтЪдаЇЙћ/ІЬg/l
|
|
ЮДМгБъ
|
МгБъ100 ІЬg/l
|
НгФЩТЪ/%
|
МгБъ200ІЬg/l
|
НгФЩТЪ/%
|
МгБъ500ІЬg/l
|
НгФЩТЪ/%
|
|
здРДЫЎ
|
0
|
96.3
|
96.3
|
188.3
|
94.2
|
478.62
|
95.6
|
|
КгЫЎ
|
0
|
93.8
|
93.8
|
196.7
|
98.4
|
482.1
|
96.4
|
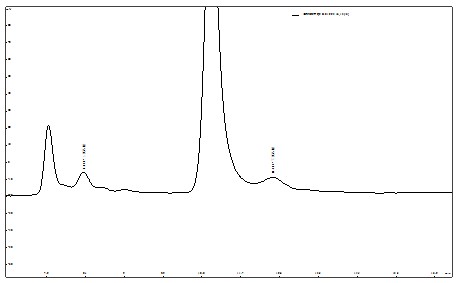
ЭМ5. здРДЫЎбљЦЗЖўТШввЫсЁЂШ§ТШввЫсМгБъЦзЭМ
3.3.4НсТл
гЩЪЕбщЪ§ОнПЩжЊ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌРызгЩЋЦзЗЈВтЪдЫЎжаЖўТШввЫсЁЂШ§ТШввЫсзМШЗЖШИп�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌМьГіХЈЖШЕЭ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌвЊСьМђЦгПьНн�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌ�ЃЌЭъШЋжЊзуЛЗБЃаавЕЙигкЫЎжЪжаЖўТШввЫсЁЂШ§ТШввЫсМьВтЕФашЧѓЁЃЁЃЁЃ�ЁЃ�ЁЃЁЃЁЃ

