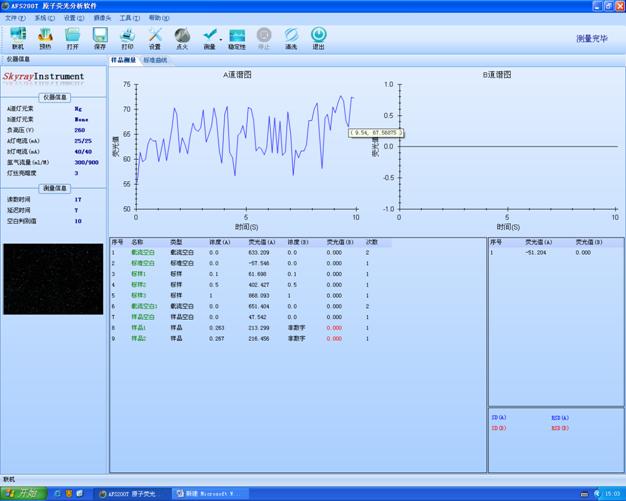
«Α ―‘
Ϋ®≤Ρ––“Β «÷–Ιζ÷ς“ΣΒΡ÷ ΝœΙΛ“Β�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘΫ®≤Ρ≤ζΤΖΑϋά®–όΫ®÷ ΝœΦΑ÷ΤΤΖΓΔΖ«Ϋπ τΩσΦΑ÷ΤΤΖΓΔΈόΜζΖ«Ϋπ τ–¬÷ Νœ»ΐ¥σΟ≈άύΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Τ’±ι”Π”Ο”Ύ–όΫ®ΓΔΨϋΙΛΓΔΜΖ±ΘΓΔΗΏ–¬ ÷“’ΙΛ“ΒΚΆ»ΥΟώ…ζ―ΡΒ»Νλ”ρ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘœ÷‘ΎΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§÷–Ιζ“―Ψ≠ «Χλœ¬…œΉν¥σΒΡ–όΫ®÷ Νœ…ζ≤ζΙζΚΆœϊΚΡΙζ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ÷ς“ΣΫ®≤Ρ≤ζΤΖΥ°ΡύΓΔΤΫΑε≤ΘΝßΓΔ–όΫ®Έά…ζ÷ ΝœΓΔ ·≤ΡΚΆ«ΫΧε÷ ΝœΒ»≤ζΝΩΕύΡξΨ”Χλœ¬ΒΎ“ΜΈΜ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘΆ§ ±Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Ϋ®≤Ρ≤ζΤΖ÷ ΝΩ“Μ÷±ΧαΗΏΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Ρή‘¥ΚΆ‘≠÷ ΝœœϊΚΡ÷πΡξœ¬ΫΒΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§÷÷÷÷–¬–ΆΫ®≤Ρ“Μ÷±”Ωœ÷Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Ϋ®≤Ρ≤ζΤΖ“Μ÷±…ΐΦΕΜΜ¥ζ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ‘ΎΩΤ―ß ÷“’Ζ…ΥΌ…ζ≥ΛΓΔΗςœν ¬“Β’τ’τ»’…œ»ΖΒ±Ϋώ…γΜαΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΥφΉ≈ΈΡΜ·ΚΆΈο÷ …ζ―ΡΥ°ΤΫΒΡ≤ΜΙήΧαΗΏΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§»ΥΟ«‘ΫΖΔΉΔ÷Ί¬Χ…ΪΩΒΫΓΒΡœϊΚΡάμΡνΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Ε‘«ι–Έ‘ΫΖΔ÷Ί ”Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§≤Δ«“‘ΫΖΔ÷Ί ”¬Χ…ΪΜΖ±ΘΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§“ρ¥ΥΕ‘Ϋ®≤Ρ≤ζΤΖΒΡΦλ≤β–η«σ‘Ϋά¥‘Ϋ¥σΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§“≤‘Ϋά¥‘Ϋ―œΩαΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§’β“Μ«–ΕΦάκ≤ΜΩΣ÷÷÷÷Φλ≤β“«Τςluck18ΙΌΆχ“«ΤςΙΪΥΨ…ζ≤ζΒΡ÷÷÷÷œΗΟήΤ Έω“«ΤςΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Ω…“‘÷ΣΉψΫ®≤Ρ––“ΒΒΡ÷ ΝΩΩΊ÷Τ–η«σΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΥθΕΧΤ Έω ±ΦδΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§≤Δ«“Τ Έω–ßΙϊΉΦ»ΖΩ…ΩΩΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§¥σ¥σΦ”ΥΌΝΥΈ“ΙζΫ®≤Ρ––“ΒΒΡ…ζ≥Λ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
“ΜΓΔ‘≠Ή”Έϋ ’Ϋ®≤Ρ––“ΒΫβΨωΦΤΜ°
“ΣΝλœ»»ίΘΚ±Ψ“ΣΝλœ»»ίΝΥ‘≠Ή”Έϋ ’Ζ®≤βΕ®Ϋ®≤Ρ÷–Ϋπ τ‘ΣΥΊΒΡœξœΗ≤β ‘“ΣΝλΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΗΟ“ΣΝλ÷ς“Σ’κΕ‘Ϋ®≤Ρ÷–Ϋπ τ‘ΣΥΊΒΡ≤βΕ®Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΨΏ”–Ω…≤ΌΉς–‘ΚΆΚήΚΟΒΊ”Π”ΟΦέ÷Β�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
1ΓΔ“«Τς

2ΓΔ―υΤΖ¥Π÷Ο≥ΆΖΘ
Ϋ®≤Ρ––“ΒΒΡ÷÷άύ”––μΕύΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§≤ν±πΒΡ―υΤΖ«Α¥Π÷Ο≥ΆΖΘ“ΣΝλ“≤≤ν±πΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Τδ÷–“ΣΝλΑϋά®Υα»ήΓΔΦν»έΒ»�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
3ΓΔ±ξΉΦ≈δ÷Τ
”Ο1000ug/ml ΒΡΒΞΈΜΥΊ±ξΉΦ÷ϋ¥φ»ή“Κ÷πΦΕ≈δ÷Τ≥…Υυ–η≈®Ε»ΒΡ ¬«ι»ή“Κ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
4ΓΔœ÷ Β―υΤΖΨΌάΐ
Ϋ®≤Ρ÷–Ά≠ΝΩΒΡ≤βΕ®
―υΤΖ¥Π÷Ο≥ΆΖΘΘΚ≥Τ»Γ0.5000g ‘―υ÷Ο”Ύ100mL…’±≠÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”5mL―ΈΥαΓΔ2ΓΪ3mLœθΥαΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§÷Ο”ΎΒγ»»Αε…œΦ”»»œϊ»ΎΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»κ5mlΗΏ¬»ΥαΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»»÷ΝΟΑ―ΧΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§»Γœ¬ά以ȧȧ�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»κ20mLΥ°Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»»œϊ»Ύ―Έάύ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘά以ȧȧ�Θ§�Θ§�Θ§�Θ§Θ§�Θ§“Τ»κ100mL»ίΝΩΤΩ÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§”ΟΥ°œΓ Ά÷ΝΩΧΕ»Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Μλ‘»�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ ‘“Κ÷–»τ”–ΧΦΜ·ΈοΓΔΙηΥαΒ»≥ΝΒμΈοΈω≥ωΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§–κΗ…Ιΐ¬Υ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
±ξΉΦ«ζœΏΒΡΜφ÷ΤΘΚΉΦ»Ζ“Τ»Γ0mLΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§0.10mLΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§0.20mLΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§0.30mLΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§0.40mLΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§0.50mLΆ≠±ξΉΦ»ή“Κ÷Ο”Ύ“ΜΉι»ίΝΩΤΩ÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Μ°Ζ÷Φ”»κ2mLœθΥαΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§”Ο»ΞάκΉ”Υ°Ε®»ίΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§‘Ύ”κ’…ΝΩ ‘“ΚœύΆ§ΧθΦΰœ¬Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§’…ΝΩœΒΝ–±ξΉΦ»ή“ΚΒΡΈϋΙβΕ»Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φθ»ΞΝψ≈®Ε»»ή“ΚΒΡΈϋΙβΕ»Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§“‘Ά≠ΒΡ≈®Ε»ΈΣΚαΉχ±ξΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΈϋΙβΕ»ΈΣΉίΉχ±ξΜφ÷Τ ¬«ι«ζœΏ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
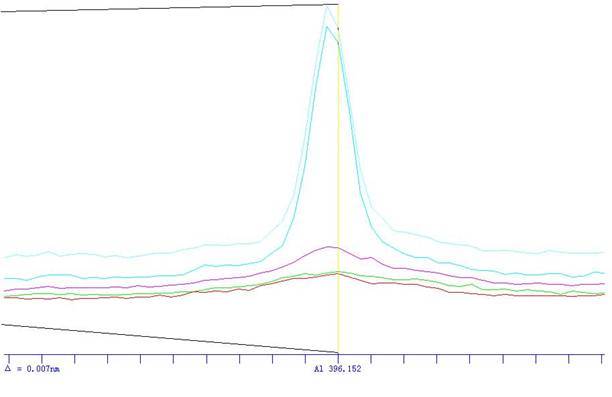
―υΤΖ≤β ‘ΘΚ”Ύ‘≠Ή”Έϋ ’ΙβΤΉ“«≤®≥Λ324.8nm¥ΠΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ Ι”ΟΩ’Τχ-““»≤Μπ―φΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§“‘Υ°ΒςΝψΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§’…ΝΩ ‘“ΚΒΡΈϋΙβΕ»Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φθ»ΞΥφΆ§ ‘ΝœΒΡΩ’»± ‘―ι»ή“ΚΒΡΈϋΙβΕ»Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§¥” ¬«ι«ζœΏ…œ≤ι≥ωœλ”ΠΒΡΆ≠≈®Ε»�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
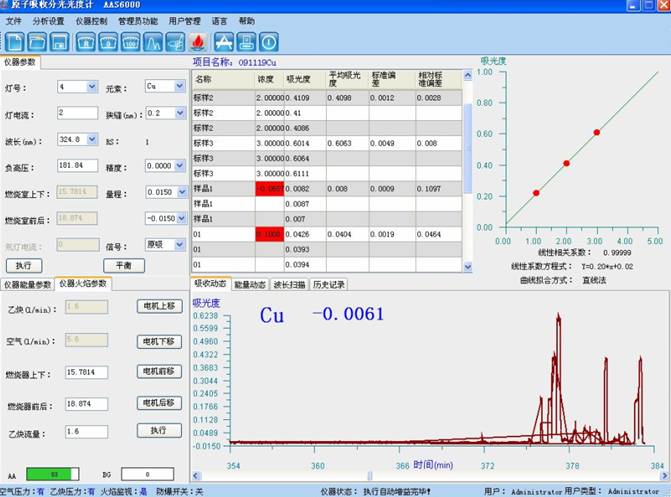
ΕΰΓΔluck18ΙΌΆχ“«ΤςICP-AESΖ®≤βΕ®Ϋ®≤Ρ÷–Ϋπ τ‘ΣΥΊΫβΨωΦΤΜ°
“ΣΝλœ»»ίΘΚ±Ψ“ΣΝλœ»»ίΝΥICPΘ≠AESΖ®≤βΕ®Ϋ®≤Ρ÷ Νœ÷–Ϋπ τ‘ΣΥΊΒΡœξœΗ≤β ‘“ΣΝλΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΗΟ“ΣΝλ÷ς“Σ’κΕ‘ΚœΫπ÷–Ϋπ τ‘ΣΥΊΚ§ΝΩΒΡ≤βΕ®Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΨΏ”–Ω…≤ΌΉς–‘ΚΆΚήΚΟΒΊ”Π”ΟΦέ÷Β�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
1. “«Τς
ICPΘ≠2000–ΆΒΞΒά…®Οη ΫICP‘≠Ή”ΖΔ…δΙβΤΉ“«(luck18ΙΌΆχ“«Τς)

2. ―υΤΖΧα»Γ
”…ΩΆΜßΧαΙ©ΨΏ”–¥ζ±μ–‘ΒΡ―υΤΖ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
3. ―υΤΖ≈δ÷Τ
ΤΨ÷Λ ‘―υ÷–‘”Κ§ΝΩΤιαΪΫΪ―υΤΖ≈δ÷Τ≥… Β±≈®Ε»,ΥαΕ»ΩΊ÷Τ‘Ύ 5ΘΞΉσ”“Φ¥Ω…�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
4. ―υΤΖ¥Π÷Ο≥ΆΖΘ
Ϋ®≤Ρ÷ ΝœΘ®≤ΘΝßΓΔΥ°ΡύΒ»Θ©”––μΕύΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§≤ν±πΒΡ―υΤΖ«Α¥Π÷Ο≥ΆΖΘ“ΣΝλ“≤≤ν±πΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§“Μ―υΤΫ≥Θ«Α¥Π÷Ο≥ΆΖΘΥυ”ΟΒΡ ‘ΦΝ”–―ΈΥαΓΔ«βΖζΥαΓΔœθΥαΓΔΗΏ¬»ΥαΓΔΥΪ―θΥ°Β»�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
«Α¥Π÷Ο≥ΆΖΘΘΚ≥Τ»Γ0.15gΉσ”“―υΤΖ”Ύ≤ΘΝß…’±≠Μρ «ΨέΥΡΖζ““œ©…’±≠÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§œ»”ΟœΓ―ΈΥα¥Π÷Ο≥ΆΖΘΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§¥ΐΖ¥”ΠΆξ»ΪΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Τχ≈ίΦθ¬ΐΚσΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”Θ®1+1Θ©œθΥαΦΧ–χœϊΫβΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§÷ΝΖ¥”ΠΆξ»ΪΘ®»τ «Ϋ®≤Ρ÷–ΙηΚ§ΝΩΫœΗΏΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΨΆ“Σ Β±Φ”«βΖζΥα¥Π÷Ο≥ΆΖΘΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§’τΖΔ“‘≥ΐ»ΞΙηΘ©Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§œϊΫβΚσΕ®»ί÷Ν100mL»ίΝΩΤΩ÷–,…œΜζ≤β ‘�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘΧΊ β «Υ°ΡύΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§“Μ―υΤΫ≥Θ“Σ”ΟΆθΥ°œϊΫβ.
5. ±ξΉΦ≈δ÷ΤΘΚ
”Ο1000ug/ml ΒΡΒΞ±ξΉΦ÷ϋ¥φ»ή“Κ÷πΦΕ≈δ÷Τ≥… 100ug/ml»ή“ΚΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§‘Ό÷πΦΕ≈δ÷Τ≥…Μλœΐ±ξΉΦ»ή“Κ10ΠΧg/mlΓΔ1ΠΧg/mlΥαΕ»ΈΣ10%(V/V)�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
6. ICP-AES ¬«ι«ζœΏ±ξΉΦ»ή“ΚΒΡ≈δ÷Τ
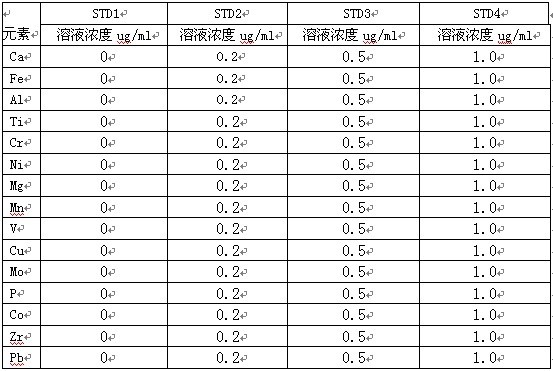
7. Τ ΈωœΏΒΡ―Γ‘ώ
ΤΨ÷ΛICP»μΦΰΤΉœΏΩβΧαΙ©Υυ”–‘ΣΥΊ≤ν±πΤΉœΏΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ Ήœ»―Γ‘ώ±Μ≤β‘ΣΥΊ―ΗΥΌΕ»ΗΏΒΡΤΉœΏΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Τδ¥ΈΥΦΝΩ≤Μ ή―υΤΖΜυΧεΦΑ±Μ≤β‘ΣΥΊœύΜΞ÷°ΦδΉΧ»≈–ΓΒΡΤΉœΏΒΡ‘≠‘ρΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§―Γ‘ώΤ ΈωœΏ»γœ¬�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
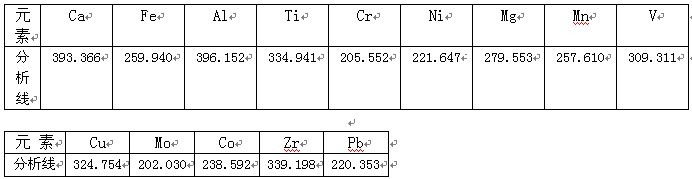
8Θ° –ßΙϊ ΐΨί
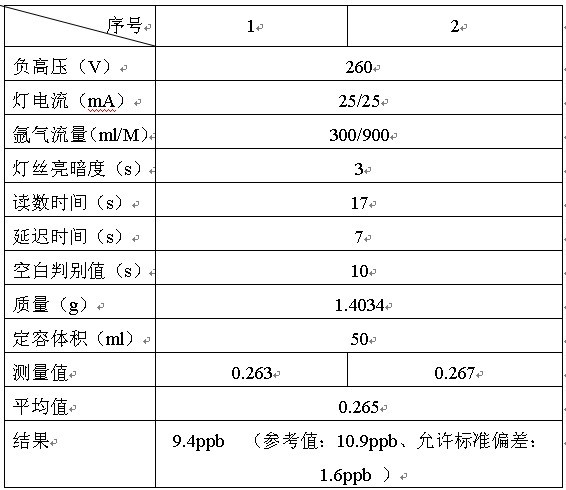
“ΣΝλΆ®Ιΐ―Γ‘ώΉνΦ― ¬«ιΧθΦΰΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Ϋ®…ηΝΥICPΘ≠2000–ΆICPΘ≠AESΖ®≤βΕ®Ϋ®≤Ρ÷–Ϋπ τ‘ΣΥΊ≤βΕ®“ΣΝλ, ΒΟ≥ω Β―ι ΐΨί�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ Β―ι÷Λ ΒΗΟ“«Τς¥σ≤Ω±πάκ“’÷Η±ξΒ÷¥οΚΆΩΩΫϋΝΥ΅κΆβΆ§άύ≤ζΤΖ ÷“’÷Η±ξ, ”…”Ύ“«Τς≤ΌΉς»μΦΰ Ι”Ο÷–ΈΡœΒΆ≥, «Ιζ»Υ Ι”ΟΗϋάϊ±ψ,’ϊΗωΤ Έωάζ≥ΧΩλΥΌΓΔ«α”·ΓΔ–ßΙϊΉΦ»ΖΓΔ”–ΚήΚΟΒΡΗΏ–≈Φέ±»Β»”≈Βψ, “‘ «ΗΟ“«Τς“≤Τ’ΦΑΒΫΗςΗωΤ ΈωΝλ≥«.
»ΐΓΔAFS200T‘ΎΫ®≤Ρ––“Β”Π”ΟΒΡΫβΨωΦΤΜ°
“ΣΝλœ»»ίΘΚ±Ψ“ΣΝλœ»»ίΝΥ‘≠Ή””ΪΙβΖ®≤βΕ®Ϋ®≤Ρ––“Β÷–Ϋπ τ‘ΣΥΊΒΡœξœΗ≤β ‘“ΣΝλΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΗΟ“ΣΝλ÷ς“Σ’κΕ‘Ϋ®≤Ρ÷–Ϋπ τ‘ΣΥΊΚ§ΝΩΒΡ≤βΕ®Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΨΏ”–Ω…≤ΌΉς–‘ΚΆΚήΚΟΒΊ”Π”ΟΦέ÷Β
1ΓΔ“«ΤςΘΚ

2ΓΔ―υΤΖ¥Π÷Ο≥ΆΖΘΦΑ‘ΣΥΊΦλ≤β
2.1AsΒΡ≤βΕ®
2.1.1ΓΓAsΒΡ±ξ“Κ…η÷Ο�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
Ι”Ο…ι(As)±ξΉΦ Ι”Ο»ή“Κ(0.1ug/ml)Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§”≈ΦΕ¥Ω―ΈΥαΘ®HCLΘ© Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Τ Έω¥ΩΝρκεΚΆΩΙΜΒ―ΣΥαΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§»ΞάκΉ”Υ°Θ®ΒγΉη¬ Γί10M ≈ΖΡΖΘ©Β» ‘ΦΝ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ

2.1.2‘ΊΝς“ΚΒΡ≈δ÷Τ
‘Ί“ΚΝςΆ®≥ȯÔϔκ±ξ“ΚΜυΧεœύ“Μ÷¬ΒΡΒ»≈®Ε»Υα»ή“ΚΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§”Ο”ΎΆΤΕ·―υΤΖ÷ΝΖ¥”ΠœΒΆ≥Φ”»κΖ¥”Π≤Δœ¥εΣ’ϊΗω“«ΤςΙή¬Ζ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
5%HCLΘ®ΧεΜΐΖ÷ ΐΘ© ΘΚΉΦ»ΖΝΩ»Γ 50ml ≈®―ΈΥαΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§”Ο»ΞάκΉ”Υ°Ε®»ί÷Ν 1000ml�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
2.1.3ΜΙ‘≠ΦΝΒΡ≈δ÷Τ
0.5%«β―θΜ·ΦΊΘ®ΡΤΘ© Θ®K(Na)OHΘ©”κ 2%≈π«βΜ·ΦΊΘ®ΡΤΘ© Θ®KΘ®NaΘ©BH4Θ©Μλœΐ»ή“Κ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
≈δ÷Τ“ΣΝλ»γœ¬ΘΚœ»ΉΦ»Ζ≥Τ»Γ 5g «β―θΜ·ΦΊΘ®ΡΤΘ©»ή”Ύ»ΞάκΉ”Υ°÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§«κ»ΖΕ®«β―θΜ·ΦΊΘ®ΡΤΘ©Άξ»Ϊœϊ»ΎΚσΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§‘ΌΉΦ»Ζ≥Τ»Γ 20g ≈π«βΜ·ΦΊΘ®ΡΤΘ©Ζ≈»κΗΟ»ή“Κ÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§”Ο»ΞάκΉ”Υ°Ε®»ίΒΫ 1000mlΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§œϊ»ΎΚσ“Γ‘»�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘΫ®“ι”Ο ±œ÷≈δΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§ΉνΚΟ≤Μ“ΣΉΓΥό…ζ―ΡΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§≈δ÷ΤΝς≥Χ≤ΜΩ…ΒΙ÷Ο�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
2.1.4…ιAs)ΒΡ―υΤΖ¥Π÷Ο≥ΆΖΘ
¥”―υΤΖ÷–≥ΤΝΩ0.20gΉσ”“”Ύ≤ΘΝß…’±≠÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»κ8ml HCl,5mlHF, 8ml HNO3�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘΦ”»»œϊΫβ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘά以ΚσΕ®»ί”Ύ100ml»ίΝΩΤΩ÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»κ5mlHClΘ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§5%ΒΡΝρΡρΚΆΩΙΜΒ―ΣΥα»ή“Κ20ml�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘΘ®Ά§ ±Ήω―υΤΖΩ’»±Θ©�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
2.1.5“«Τς≤ΌΉςΧθΦΰΘΚ
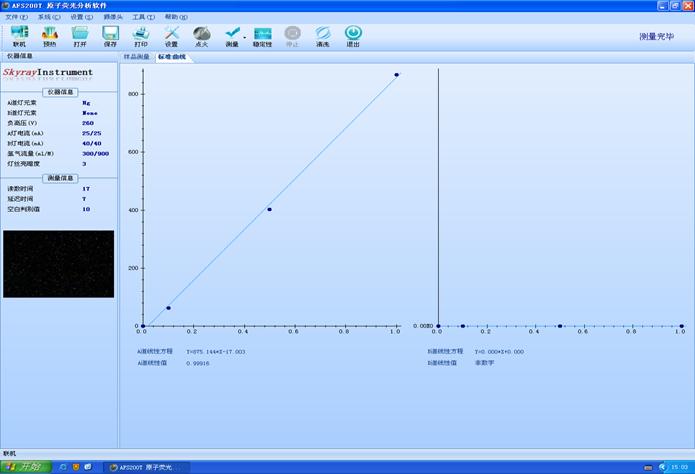
ΗΚΗΏ―ΙΘ®VΘ©:270�ΘΜ�ΘΜΘΜ�ΘΜΘΜ�ΘΜΒΤΒγΝςΘ®mAΘ©:40/40�ΘΜ�ΘΜΘΜ�ΘΜΘΜ�ΘΜκ≤ΤχΝςΝΩΘ®ml/MΘ©:300/800
ΒΤΥΩΝΝΑΒΕ»:3 �ΘΜ�ΘΜΘΜ�ΘΜΘΜ�ΘΜΕΝ ΐ ±Φδ:12s�ΘΜ�ΘΜΘΜ�ΘΜΘΜ�ΘΜ―”≥Ό ±Φδ:2s�ΘΜ�ΘΜΘΜ�ΘΜΘΜ�ΘΜΩ’»±≈–±π÷Β:2
3ΓΔ”Π”Ο Βάΐ
3.1.ΆΩΝœ÷–≤βΙ·
«Α¥Π÷Ο≥ΆΖΘΘΚ¥”―υΤΖ÷–≥ΤΝΩ1.4034g”Ύ≤ΘΝß…’±≠÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»κ8ml HNO3ΚΆ2mlHClO4Φ”»»œϊ»ΎΘ®œϊΫβΈ¬Ε»350ΓφΉσ”“Θ©�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘά以ΚσΕ®»ί”Ύ50ml»ίΝΩΤΩ÷–Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§Φ”»κ1ml HNO3Θ§Θ§�Θ§�Θ§�Θ§�Θ§Θ§�Θ§5%ΒΡ÷ΊΗθΥαΦΊ1ml�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘΘ®Ά§ ±Ήω―υΤΖΩ’»±Θ©Θ®ΜΙ‘≠ΦΝΦ”0.5%Χζ«ηΜ·ΦΊΘ©
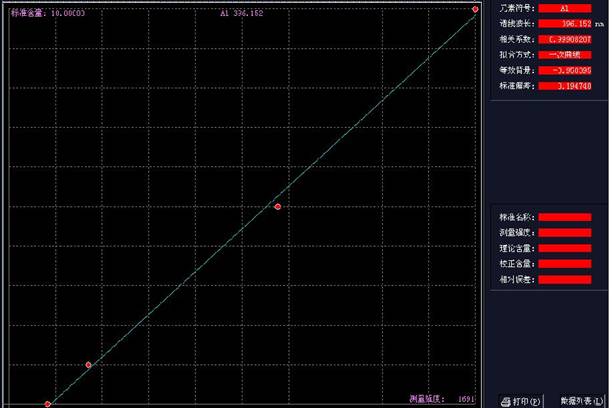
±ξΉΦ»ή“Κ…η÷Ο≈®Ε»ΘΚ0.1ppb 0.5ppb 1ppbΉς«ζœΏ�ΓΘ�ΓΘ�ΓΘ�ΓΘΓΘΓΘΓΘ
Θ®AΒάœΏ–‘ΖΫ≥ΧΘΚY=875.144*X-17.003Θ©�ΘΜ�ΘΜΘΜ�ΘΜΘΜ�ΘΜΘ®AΒάœΏ–‘÷ΒΘΚ0.99916Θ©ΦϊΆΦ1
“«Τς≤ΌΉςΧθΦΰΘΚ
ΆΦ1